VictorBorun wrote: ↑Mon Sep 06, 2021 10:03 pm
johnnydeep wrote: ↑Mon Sep 06, 2021 6:15 pm
That'll do:
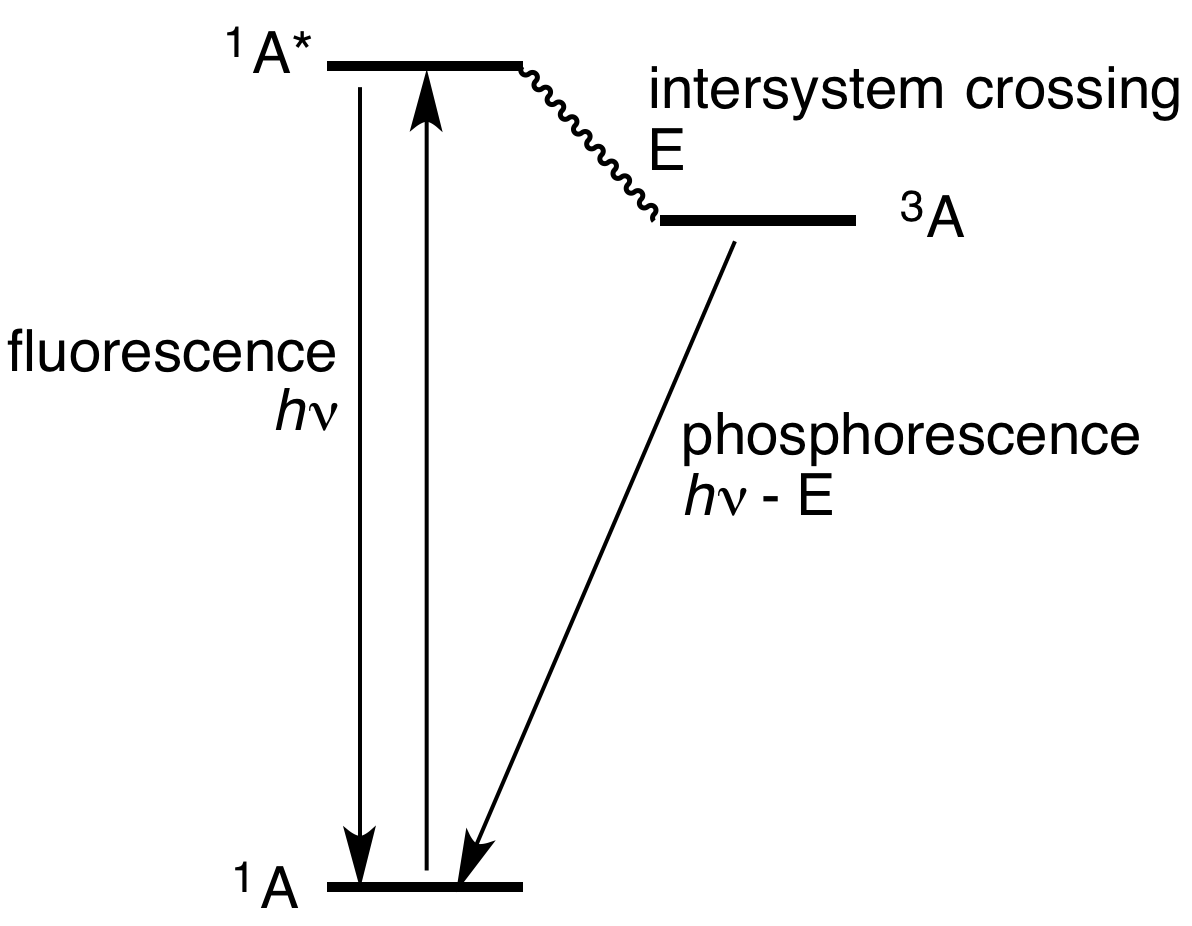
Airglow’s subtle radiance arises from excitation of a different kind [than that of auroras]. Ultraviolet light from the daytime sun ionizes or knocks electrons off of oxygen and nitrogen atoms and molecules; at night the electrons recombine with their host atoms, releasing energy as light of different colors including green, red, yellow and blue. The brightest emission, the one responsible for creating the green streaks and bands visible from the ground and orbit, stems from excited oxygen atoms beaming light at 557.7 nanometers, smack in the middle of the yellow-green ██ parcel of spectrum where our eyes are most sensitive.
Where is that spooky
nebulium ██ glow at 500.7 nm and 495.9 nm ?
Is not our Sun good enough for pumping oxigen to O²⁺, or OIII ?
Don't know. I guess either emission at those wavelengths is happen but just not brightly enough to see here, or maybe some mechanism prevents those excited states from occurring in the first place.
And for those wondering, like I was, about "nebulium", here's
https://en.wikipedia.org/wiki/Nebulium :
In the early days of telescopic astronomy, the word nebula was used to describe any fuzzy patch of light that did not look like a star. Many of these, such as the Andromeda Nebula, had spectra that looked like stellar spectra, and these turned out to be galaxies. Others, such as the Cat's Eye Nebula, had very different spectra. When William Huggins looked at the Cat's Eye, he found no continuous spectrum like that seen in the Sun, but just a few strong emission lines. The two green lines at 495.9 nm and 500.7 nm were the strongest.[1] These lines did not correspond to any known elements on Earth. The fact that helium had been identified by the emission lines in the Sun in 1868, and had then also been found on Earth in 1895, encouraged astronomers to suggest that the lines were due to a new element. The name nebulium (occasionally nebulum or nephelium) was first mentioned by Margaret Lindsay Huggins in a short communication in 1898, although it is stated that her husband occasionally used the term before.[2]
And from
https://en.wikipedia.org/wiki/Doubly_ionized_oxygen :
In astronomy and atomic physics, doubly ionized oxygen is the ion O2+ (O III in spectroscopic notation). Its emission forbidden lines in the visible spectrum fall primarily at the wavelength 500.7 nm, and secondarily at 495.9 nm. Before spectra of oxygen ions became known, these lines once led to a spurious identification of the substance as a new chemical element. Concentrated levels of O III are found in diffuse and planetary nebulae. Consequently, narrow band-pass filters that isolate the 500.7 nm and 495.9 nm wavelengths of light, that correspond to green-turquoise-cyan spectral colors, are useful in observing these objects, causing them to appear at higher contrast against the filtered and consequently blacker background of space (and possibly light-polluted terrestrial atmosphere) where the frequencies of [O III] are much less pronounced.
These emission lines were first discovered in the spectra of planetary nebulae in the 1860s. At that time, they were thought to be due to a new element which was named nebulium. In 1927, Ira Sprague Bowen published the current explanation identifying their source as doubly ionized oxygen.[1]
PS - leave it to Victor to find a way to write O²⁺ without using the [ sup ] [ / sup ] tags like I did in the wikipedia quote

 Firefly Milky Way over Russia
Firefly Milky Way over Russia